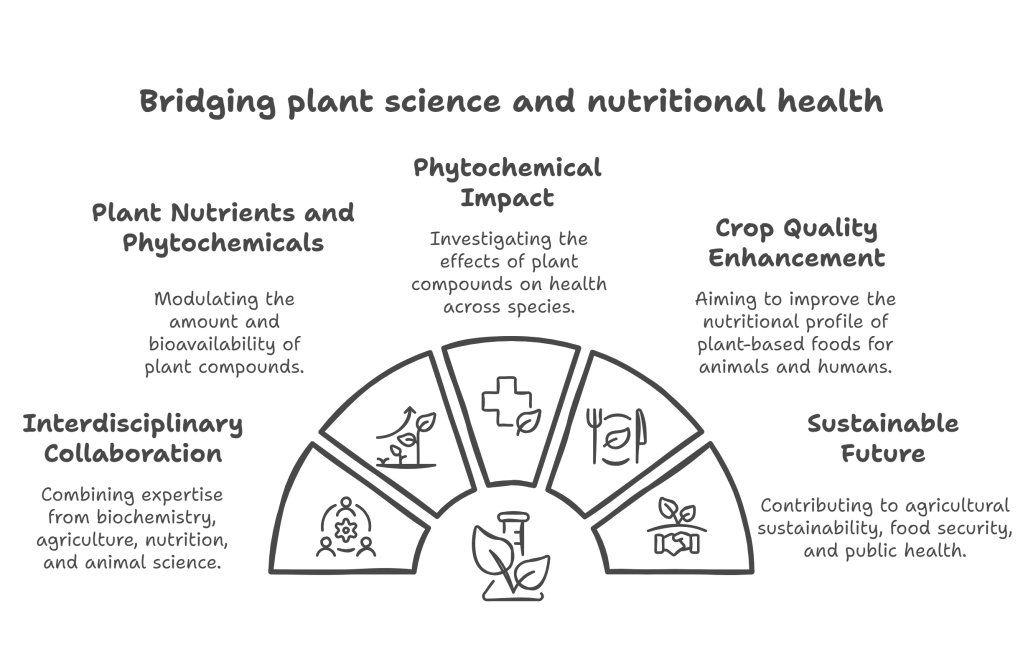
Bridging plant science and nutritional health, the Lima Lab’s research investigates the multifaceted impacts of plant compounds on plant, animal, and human health. Our research explores micronutrient bioavailability, environmental influences on phytochemical composition, and the intricate relationship between nutrition and health. We currently focus on the quantification of phytoestrogens in various sources (legume grains, forages, and animal tissues), and the examination of their impact on animal health. This includes studying phytoestrogens’ effects on dairy cow health and milk quality, as well as exploring the potential of isoflavones as dietary supplements for non-ruminant animals. Additionally, we use animal models to elucidate the underlying mechanisms of action of these phytochemicals in disease prevention.
The Lima Lab employs a combination of fundamental and applied approaches, collaborating across diverse disciplines such as biochemistry, agriculture, nutrition, and animal science. Our ultimate goal is to enhance crop quality and improve the nutritional profile of plant-based foods for both animal and human consumption. This interdisciplinary approach contributes to a better understanding of plant-derived compounds, potentially informing future developments in sustainable agriculture and evidence-based nutrition. By elucidating the complex interactions between plants, animals, and human health, our research aims to address critical challenges in food security, agricultural sustainability, and public health.
Projects
Potential Benefits of Soybean Isoflavones to Swine Health
PI: Lima co-pi: El-kadi
2023-2025 united soybean board
Non-technical summary: Isoflavones can contribute to improving animal growth and health, and reduce antibiotic use in livestock operations. However, there is not a clear understanding of how isoflavones benefit pigs’ growth and health. In this project, we will conduct a feeding trial supplementing pig diets with soybean isoflavones to investigate: 1) isoflavone exposure and accumulation in nursery pigs, and 2) the effect of isoflavone supplementation on nursery pigs’ growth and health indicators. Our results will provide useful information on the use of soybean isoflavones as growth promoters.
Fenics: fe-Nutrition Improvement with chitosan & sensors
PI: Lima co-pi: ali
2022-2023 Translational plant sciences center
Non-technical summary: Iron deficiency chlorosis (IDC) is an important agronomical problem in soybean production. Usually, iron is not limited in soils, but its bioavailability for plants is an issue. Finding sustainable products and practices to ameliorate IDC is a priority. We propose to study the role of a natural product, chitosan, in improving IDC, and develop an iron sensor prototype. The development of these products could considerably advance the smart and environmentally sustainable management of IDC.
Evaluating Soybean Varieties for Isoflavone Health Bioactive Components
PI: Lima
2022-2023 Virginia Soybean board
Non-technical summary: Soybeans and soybean products are rich in a class of bioactive compounds named isoflavones. Isoflavones, have been described as beneficial to human health and have been attracting the attention of livestock industries due to their potential role in increasing growth and improving animal health. There is an opportunity to improve soybean isoflavone profile that would profit soybean producers. In this project, we will: 1) characterize the isoflavone content of germplasm material, as well as the isoflavone content of selected soybean lines bred for use in Virginia, and 2) investigate the association of isoflavone content with other important agronomic traits. These results will contribute to the long-term goal of developing soybeans with enhanced isoflavone quality, or value-added specialty soybeans that can be used as a source of health bioactive compounds.
PHYTOCHEMICALS: IMPROVING PLANT HEALTH AND NUTRIENT PROFILE FOR HUMAN CONSUMPTION
PI: Lima
2019-2021 USDA NIFA, Hatch project 1020314
Non-technical summary: A high daily intake of fruits and vegetables is associated with prevention of costly and debilitating chronic diseases. Despite all the evidence and the public health campaigns promoting fruit and vegetable consumption, the intake of these foods is seriously below the recommended amounts. To address this problem, improvement of health beneficial nutrients in fruits and vegetables, and reduction of barriers to fruit and vegetable consumption, such as cost and pesticide residue, are important actions. Grapes are a fruit rich in health beneficial phytochemicals, and therefore an important crop to target when aiming at improving populations’ fruit consumption. The recent availability of cold-hardy grape varieties allowed the successful cultivation of these fruits in cold climates, including in New England. The results from this study are expected to provide information on the association of training system (method used to hold fruiting canes in a support structure/trellis) with incidence or severity of common grapevine diseases. Furthermore, this work is expected to inform if training systems can be used as a sustainable cultural practice that allows modulating fruit chemical composition (in a way to enhance grapes’ resistance to disease, while improving the health-beneficial properties of the fruits).
A MULTIREGIONAL APPROACH TO BALANCING MILK AND FORAGE QUALITY TRADEOFFS IN ORGANIC DAIRIES FEEDING HIGH-LEGUME DIETS
PI: Brito Co-PIs: Smith, Lima, Tsang, Heins, Romero, Hadrich
2020-2024 USDA NIFA Organic Agriculture Research and Extension Initiative
Non-technical summary: The project long-term goal is to enable farmers to adopt practices that enhance the persistence of legumes in pastures and hayfields and to produce forages with high nutritive value in order to minimize off-farm grain costs, produce milk with enriched fat and protein content, and capitalize on industry premiums. Our research and educational program will synergize the efforts of two premier land-grant organic dairy research farms (University of New Hampshire and University of Minnesota) and the University of Maine to provide farmers with information and actionable strategies for managing mixed grass-legume stands to optimize legume proportion, persistence, and quality. At the same time, we will build a baseline dataset on quality components and phytoestrogen concentrations in forage legumes and milk that will guide future research and extension. Our objectives were developed in consultation with organic dairy stakeholders in our respective regions as part of our recently completed OREI planning grant needs assessment. Team overarching objectives are to (Objective 1) Improve perennial legume abundance and persistence in mixed forage swards utilized for grazing and conserved feed; (Objective 2) Improve forage energy:protein balance, nutritive value, and milk quality components through interseeding practices and selected legume-grass mixtures; (Objective 3) Quantify and mitigate potential impacts of phytoestrogens from legumes on herd reproductive health by evaluating high- vs. low-phytoestrogenic species and how forage conservation methods affect the phytoestrogen contents of hay and silage; and (Objective 4) Develop and deliver educational programing to organic dairy farmers on advanced forage legume management aimed at increasing milk production and quality through webinars, workshops, and on-farm demonstrations. Project impact will be evaluated by The Organic Center and our Research and Extension Advisory Board.
DIVERSIFYING THE MAPLE SYRUP INDUSTRY TO ENHANCE SOCIOECOLOGICAL RESILIENCE & SUSTAINABILITY
PI: Asbjornsen Participant: Lima
2021 UNH Collaborative Research Excellence (CoRE) Pilot Research Partnership
Summary: The maple syrup industry is a quintessential feature of New England’s landscape, economy, culture, and environment. However, because the industry relies on a single species (sugar maple) that is known to be vulnerable to many stressors—including climate change and various pests and pathogens—its resilience is remarkably low. This interdisciplinary collaborative initiative will explore the potential for diversifying the maple syrup industry by utilizing other non-maple tree species from the NE region for syrup production. By bringing together scientists from the ecological, pharmacological, nutritional, and economic disciplines along with industry and extension partners, we will address the following three objectives: (1) assess the ecology and productivity of winter sap flow in several promising (non-maple) tree species; (2) evaluate the chemical properties and potential nutritional impact of the novel tree saps and syrups from these species; (3) determine the economic and market potential of these novel syrups. Our long-term goal is to develop science-based management guidelines for a diversified and resilient syrup industry based on the demonstrated ecological, health, nutritional, and economic potential of novel tree saps and syrups.

You must be logged in to post a comment.